Dr Gloria Gonzalez-Aseguinolaza, Chief Scientific Officer of Vivet Therapeutics Receives the Rosalind Franklin Society Special Award in Science

Paris, France, July 16, 2024 – Vivet Therapeutics (“Vivet”), a clinical stage biotech company developing novel and long-lasting gene therapies for rare inherited liver metabolic disorders, is delighted to announce that its Co-Founder & Chief Scientific Officer, Dr Gloria Gonzalez-Aseguinolaza, PhD, has been honored with the Rosalind Franklin Society (RFS) Special Award in Science given […]
Characteristics of patients with Wilson Disease in the United States: An insurance claims database study
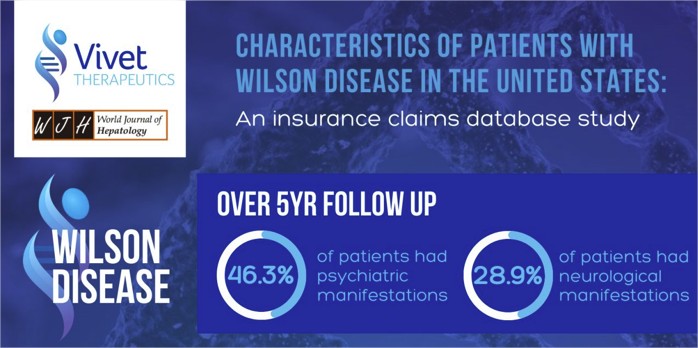
We are delighted to share our findings in a peer reviewed paper published in the World Journal of Hepatology, titled ‘Characteristics of patients with Wilson Disease in the United States: An insurance claims database study.’
Vivet Therapeutics presents interim data on its Phase 1/2 GATEWAY trial for the Treatment of Wilson Disease at EASL Congress 2024

Vivet Therapeutics Presents Interim Data from its Phase 1/2 GATEWAY Trial for the Treatment of Wilson Disease at EASL Congress 2024 VTX-801 increased ceruloplasmin ferroxidase activity and improved liver histology
Study on Treatment of Wilson Disease in Japan Published in Japanese Journal of Gastroenterology and Hepatology
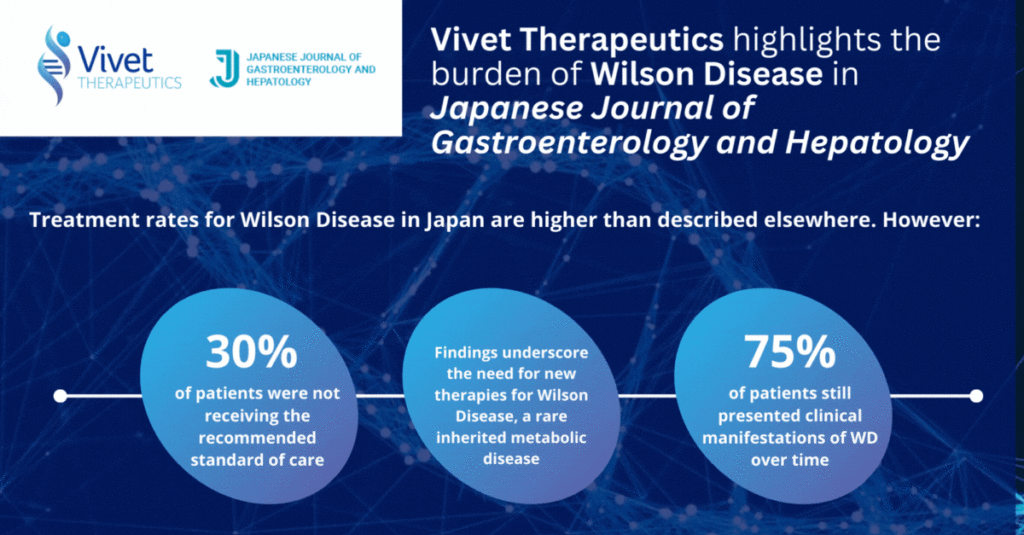
Paris, France, May 30, 2024 – Vivet Therapeutics (“Vivet”), a clinical stage biotech company developing novel and long-lasting gene therapies for rare inherited liver metabolic disorders,
Vivet Therapeutics Doses First Patient in Cohort 2 in Phase 1/2 GATEWAY Clinical Trial for Treatment of Wilson Disease

Vivet Therapeutics Doses First Patient in Cohort 2 in Phase 1/2 GATEWAY Clinical Trial for Treatment of Wilson Disease
Rare Disease Day – February 29

To mark #RareDiseaseDay on 29 February, our CEO Jean-Philippe Combal, spoke to pharmaphorum
Vivet Therapeutics receives EUR 4.9 million to advance development of a gene therapy

PRESS RELEASE Vivet Therapeutics receives EUR 4.9 million to advance development of a gene therapy for the treatment of cerebrotendinous xanthomatosis
Vivet Therapeutics Announces Presentations at Upcoming European Society of Gene and Cell Therapy (ESGCT) 2023 Annual Congress

Paris, France, October 23, 2023 (GLOBE NEWSWIRE) – Vivet Therapeutics (“Vivet”), a clinical- stage biotechnology company
Vivet Therapeutics awarded with the Prix Galien MedStartup in New York

Vivet Therapeutics is very pleased and proud to have won the Prix Galien MedStartup Award for “Best Collaboration For the Developing Or Underserved Populations Worldwide”.
GATEWAY clinical trial for Wilson Disease has now its website

Wilson Disease is a rare, progressive genetic disorder that causes excess copper to be stored in the body.

